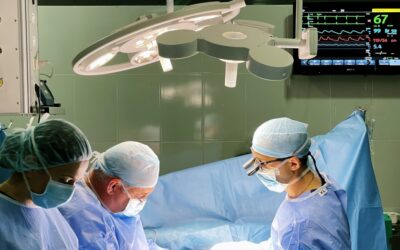
Welcome to AtriAN
AtriAN Medical

Mission & Vision
Resolving Atrial Fibrillation at its origin

TECHNOLOGY
A Disruptive Approach to Rhythm restoration
THE DISRUPTION
AtriAN’s Business Pathway

Growing Market

Robust and Innovative Intellectual Property (IP)

Unique Solution

Funding in Place

Unmet Need

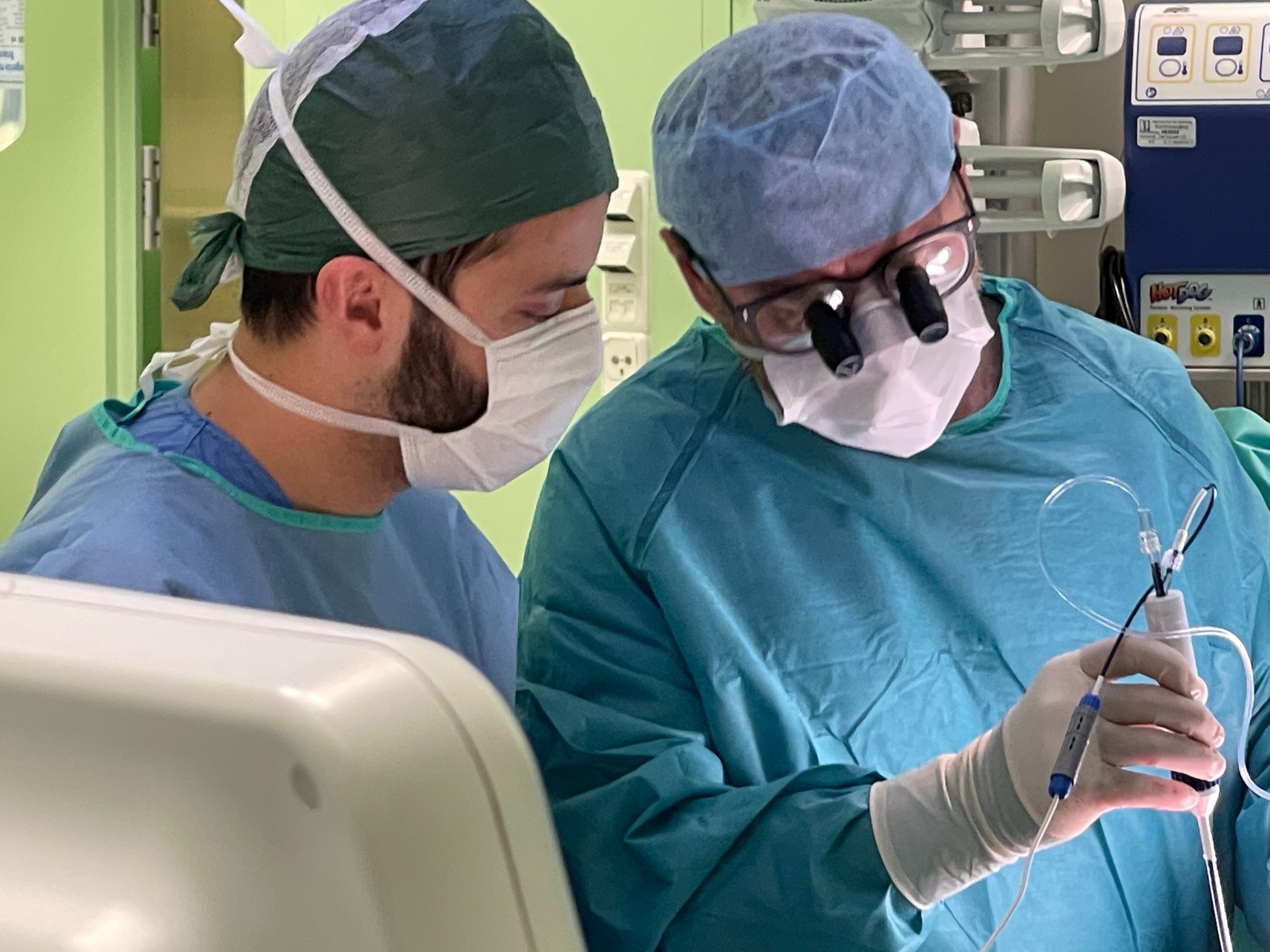
First In Human (FIH) Trials
Clinical Trials
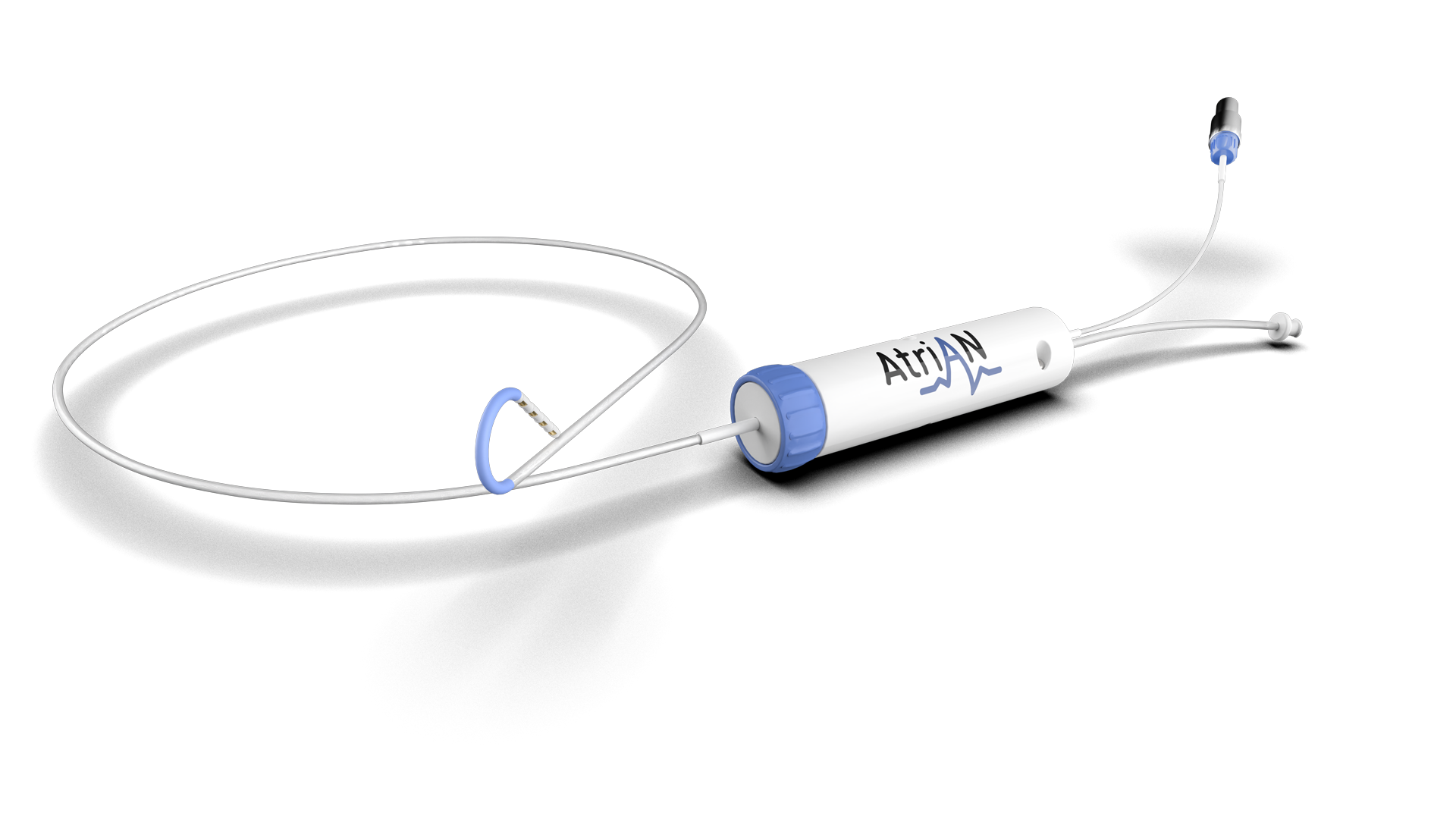
Pulsed Field Ablation (PFA)
Autonomics Remodeling
Sub-Xiphoid Approach

GENTLE, NON-THERMAL ENERGY
Pulsed Field Cardioneuroablation
Selectively targets and disables the GPs
Gently spares the myocardium (heart muscle).

Gentle
As a result, the other tissues, such as healthy heart muscle, are spared.
Non-Thermal
Long-Term

Unique, IP-Protected Catheters
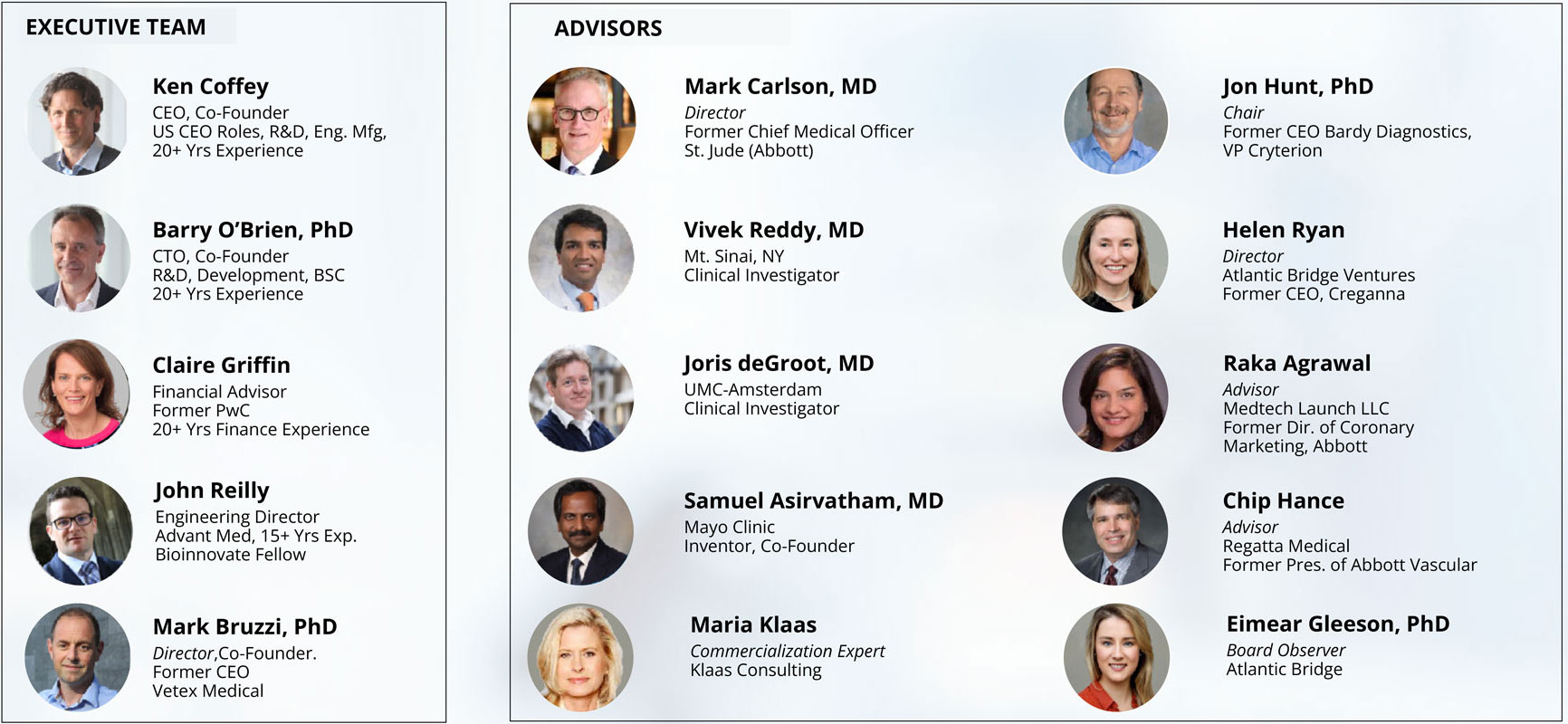
Our Team
AtriAN team members
Publications

Targeted ablation of epicardial ganglionated plexi during cardiac surgery with pulsed field electroporation (NEURAL AF)
DR Musikantow, VY Reddy, I Skalsky et al
J Interv Card Electrophysiol 2023.
Cardioneuroablation Using Epicardial Pulsed Field Ablation for the Treatment of Atrial Fibrillation
B O’Brien, J Reilly, K Coffey et al
J. Cardiovasc. Dev. Dis. 2023.
Novel Percutaneous Epicardial Autonomic Modulation in the Canine for Atrial Fibrillation: Results of an Efficacy and Safety Study
Pre-Clinical
M Madhavan, KL Venkatachalam, MJ Swale et al
Pacing Clin Electrophysiol 2016.
This was the first screening study at Mayo Clinic to assess the best energy source for ablation of ganglionated plexi, while minimizing myocardial damage. Following selection of pulsed field ablation (PFA) from the screening work, procedural feasibility was also demonstrated in both open-chest and sub-xiphoid models, using early prototype devices.
Electroporation of epicardial autonomic ganglia: Safety and efficacy in medium‐term canine models
Pre-Clinical
D Padmanabhan, N Naksuk, AK Killu et al
J Cardiovasc Electrophysiol 2019.
This study was performed using sub-xiphoid access with acute measurements showing that ganglionated plexi ablation produced an extension in the atrial effective refractory period (AERP). Extensive histology evaluations at four month follow-up demonstrated ablation of ganglia structures with sparing of the myocardium. The animals were resistant to AF induction at four months.
Open-chest Pulsed Electric Field Ablation of Cardiac Ganglionated Plexi in Acute Canine Models
Pre-Clinical
M van Zyl, M Khabsa, JA Tri et al
J Innov Cardiac Rhythm Manage 2022.
This open-chest study was performed as full system validation before progressing to first-in-human open-chest clinical trials. It’s utilized AtriAN’s cardiac-customized pulsed field generator and devices designed specifically for open-chest epicardial treatment. Acute extensions in atrial effective refractory period (AERP) were demonstrated as well as evidence of reversible electroporation in myocardial tissue.
Safety and Feasibility of Epicardial Percutaneous Access and Maneuverability With a Novel Ablation Catheter for the Treatment of Atrial Fibrillation in a Porcine Model
Pre-Clinical
P Buszman, K Lukasik, S Deane et al
J Am Coll Cardiol 2021 (Supplement).
This was a first feasibility of epicardial access using the initial prototype of a next generation catheter system designed for sub-xiphoid access and with bipolar ablation capability. Access to the pericardial space was demonstrated with conventional needle technology and the catheter demonstrated promising tracking and navigation performance.
Epicardial Pulsed Field Ablation of Ganglionated Plexi: Computational and Pre-Clinical Evaluation of a Bipolar Sub-Xiphoid Catheter for the Treatment of Atrial Fibrillation
B O’Brien, J Reilly, K Coffey et al
Bioengineering 2024.
Establishing Irreversible Electroporation Electric Field Potential Threshold in A Suspension In Vitro Model for Cardiac and Neuronal Cells
In-Vitro Studies
S Avazzadeh, B O’Brien, K Coffey et al
Journal of Clinical Medicine 2021
This in-vitro cellular study explored the electroporation thresholds of neurons and cardiomyocytes in suspension. This is the first published data showing that the thresholds for both cell types are similar; the data for neuron cell bodies being much lower than published data on axons. This provides initial data towards understanding the mechanism of selective ablation of ganglionated plexi.
Establishing electroporation thresholds for targeted cell specific cardiac ablation in a 2D culture model
In-Vitro Studies
S Avazzadeh, MH Dehkordi, P Owens et al
J Cardiovasc Electrophysiol 2022.
This study of in-vitro electroporation assessed cultured layers of cardiomyocytes and neurons and showed that neurons have a slightly lower threshold compared to cardiomyocytes. In addition neurons showed continued cell death up to 24 hours after treatment, while cardiomyocytes trended towards low levels of recovery. This insight provides further understanding of how ganglionated plexi may be ablated and myocytes spared in an in vivo scenario.
Full torso and limited-domain computer models for epicardial pulsed electric field ablation
Electric Field Models
A González-Suárez, RM Irastorza, S Deane et al.
Computer Methods and Programs in Biomedicine 2022.
This is a fundamental electric field model study aiming to demonstrate the applicability of a model geometry around the treatment zone in an epicardial configuration. This monopolar model showed how the peak electric field strengths are located very close to the catheter electrodes and drop off very rapidly beyond this. The field strength levels in adjacent organs were exceptionally low, verifying the absence of collateral tissue damaged observed in pre-clinical studies.
Pulsed Electric Field Ablation of Epicardial Autonomic Ganglia: Computer Analysis of Monopolar Electric Field across the Tissues Involved
Electric Field Models
A González-Suárez, B O’Brien, M O’Halloran et al
Bioengineering 2022
This electric field modelling study assessed the field strength distribution in the epicardial fat pads (where the ganglionated plexi are located) and the underlying myocardium, when monopolar pulses are applied. The field strength tends to concentrate within the fat layer, with a substantial drop off into the myocardium. This effect further explains the observed selectivity of ganglia ablation when the treatment is performed epicardially.
In Silico Modelling to Assess the Electrical and Thermal Disturbance Provoked by a Metal Intracoronary Stent during Epicardial Pulsed Electric Field Ablation
A González-Suárez, J Perez, B O’Brien et al
J. Cardiovasc. Dev. Dis. 2022
News

contact us
Get In Touch
Address
Unit 1,
BIC Centre
Upper Newcastle
Galway Ireland
H91E79C
Visit
Visit us in Galway
Unit 204 (Billing), H91W60E
Atrian
Upper Newcastle
Galway, Ireland
H91E79C